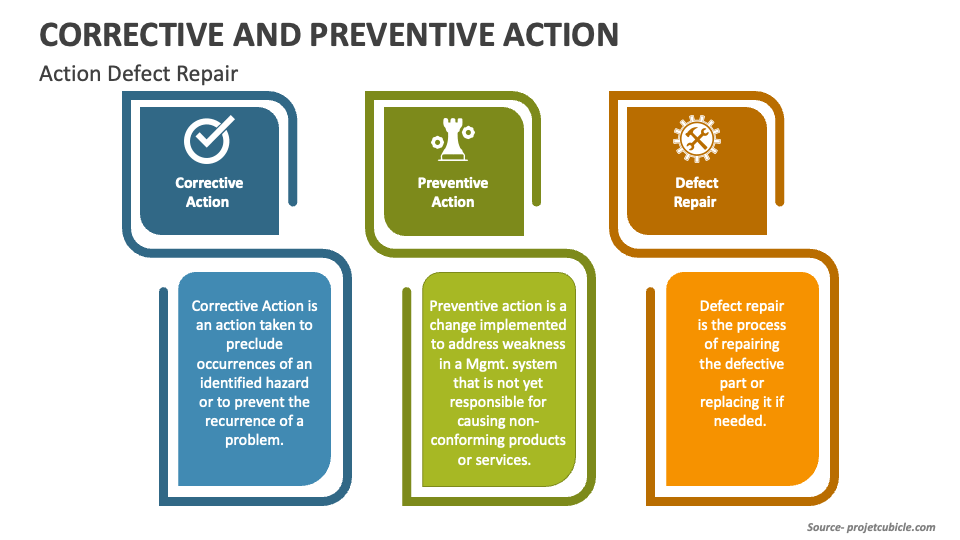
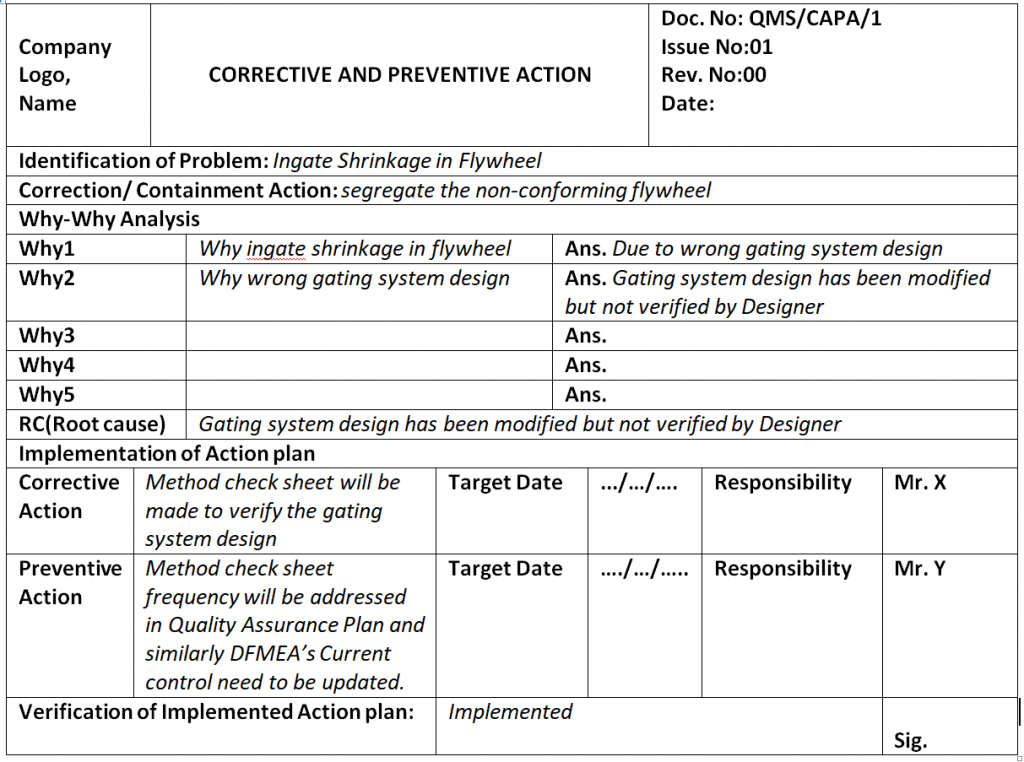
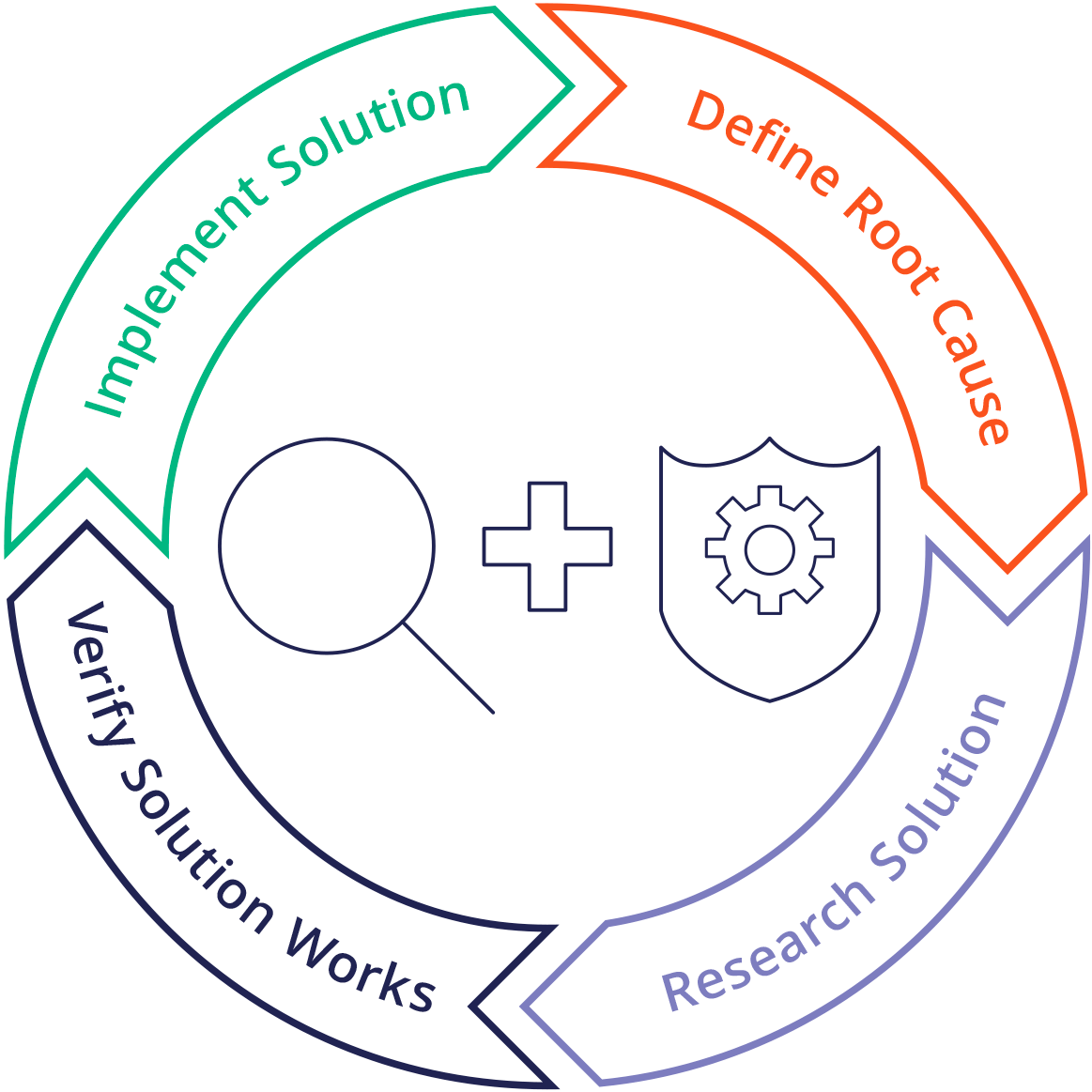
Corrective And Preventive Action System - Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective and preventive actions (capa) inspectional objectives.
Corrective and preventive actions (capa) inspectional objectives. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,.
Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Capa methodology is useful where corrective actions and preventive actions are incorporated. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Food and drug administration (fda), corrective and preventive. Corrective and preventive actions (capa) inspectional objectives.
Corrective and Preventive Action Procedure
Purpose of the corrective and preventive action subsystem •to communicate corrective and. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective and preventive actions (capa) inspectional objectives. Food and drug administration (fda), corrective and preventive.
Corrective and Preventive Action PowerPoint Presentation Slides PPT
Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Corrective and preventive actions (capa) inspectional objectives. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,.
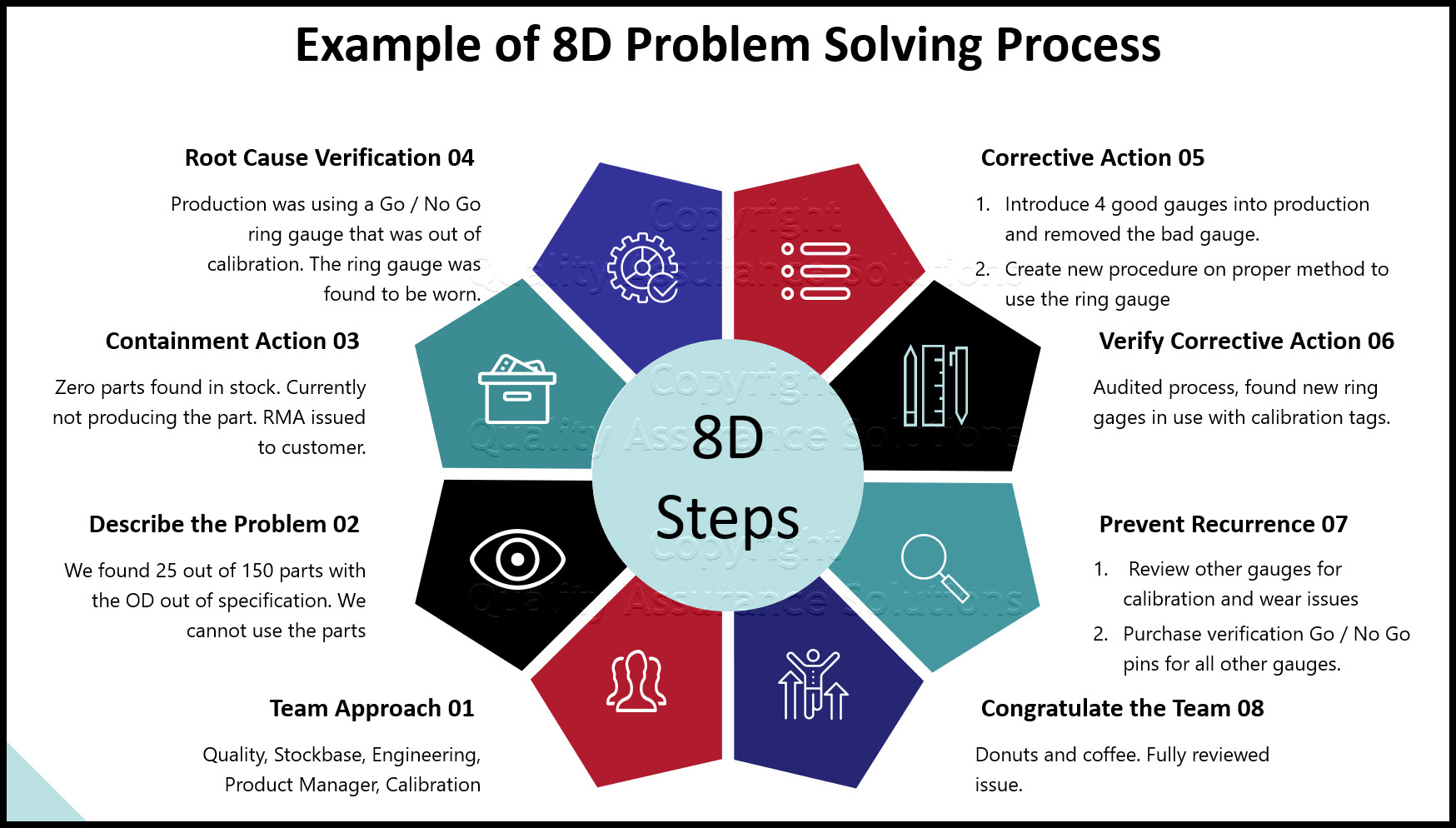
CAPA Corrective and Preventive Action. Graphic by DGStudio
Capa methodology is useful where corrective actions and preventive actions are incorporated. Food and drug administration (fda), corrective and preventive. Corrective and preventive actions (capa) inspectional objectives. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Purpose of the corrective and preventive action subsystem •to communicate corrective and.
CAPA Systems 5 Essential Elements CAPA Software Arena
Corrective and preventive actions (capa) inspectional objectives. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,.
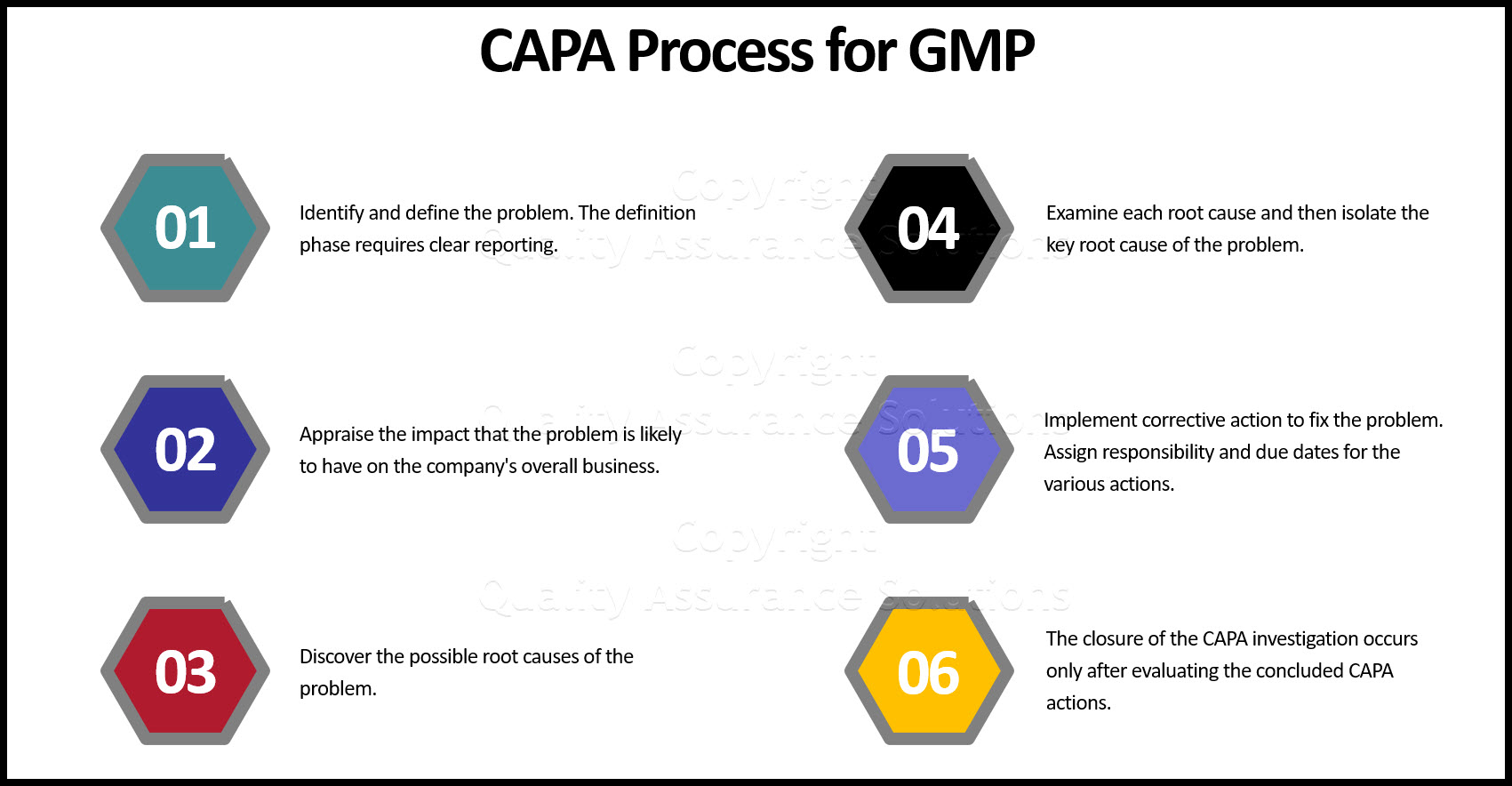
Preventive Corrective Action With 6 Steps
Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Food and drug administration (fda), corrective and preventive. Corrective and preventive actions (capa) inspectional objectives. Capa methodology is useful where corrective actions and preventive actions are incorporated.
Preventive Corrective Action With 6 Steps
Corrective and preventive actions (capa) inspectional objectives. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Purpose of the corrective and preventive action subsystem •to communicate corrective and.
Preventive Corrective Action With 6 Steps
Purpose of the corrective and preventive action subsystem •to communicate corrective and. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Corrective and preventive actions (capa) inspectional objectives. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated.
Corrective Action Preventive Action Template
Corrective and preventive actions (capa) inspectional objectives. Food and drug administration (fda), corrective and preventive. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Purpose of the corrective and preventive action subsystem •to communicate corrective and.
Corrective and Preventive Action (CAPA) Definition, Purpose, and
Corrective and preventive actions (capa) inspectional objectives. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Food and drug administration (fda), corrective and preventive.
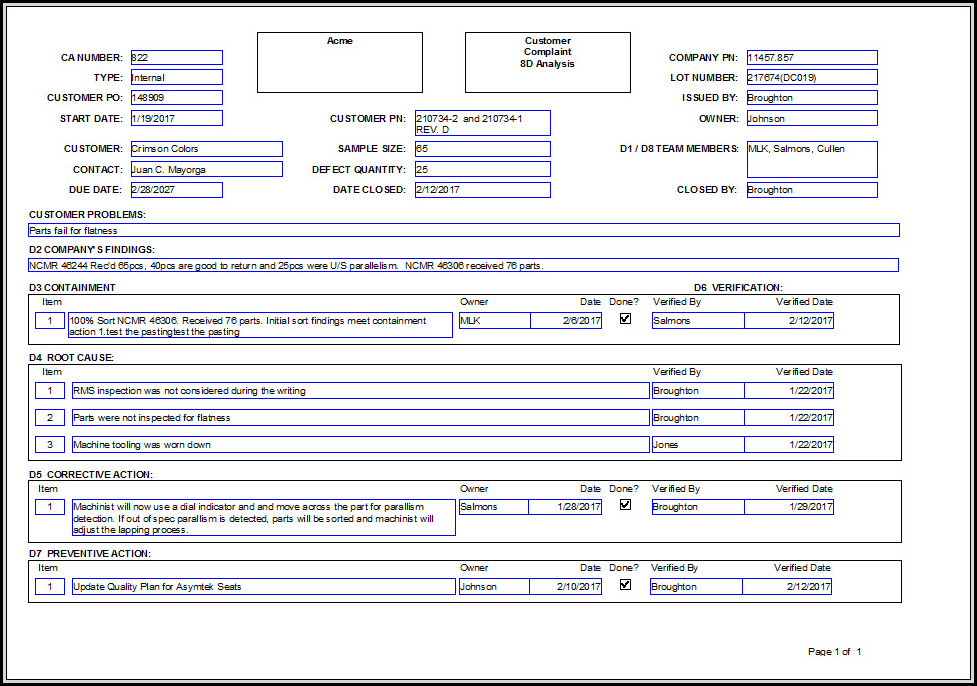
Corrective Action and Preventive Action PDF Personal Protective
Food and drug administration (fda), corrective and preventive. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Purpose of the corrective and preventive action subsystem •to communicate corrective and. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective and preventive actions (capa) inspectional objectives.
Purpose Of The Corrective And Preventive Action Subsystem •To Communicate Corrective And.
Food and drug administration (fda), corrective and preventive. Corrective actions (ca) take steps to fix the cause of a problem after the problem has occurred,. Capa methodology is useful where corrective actions and preventive actions are incorporated. Corrective and preventive actions (capa) inspectional objectives.