Differentiate Between Octahedral Fe 2 And Fe 3 In Fe24O32 - This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I understand that fe(ii) has 6. Start with an oh energy. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions.
This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Start with an oh energy. I understand that fe(ii) has 6. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions.
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. Start with an oh energy. I understand that fe(ii) has 6.
Plot of Li versus octahedral Fe 2þ cations for the magmatic grains. All
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Start with an oh energy. I.
Solved If an octahedral Fe*2 complex is which
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I understand that fe(ii) has 6..
What is the standard reduction potential (E°) Fe3+ Fe ? Given that Fe2
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I understand that fe(ii) has 6..
Why is Fe2+ more easily oxidized to Fe3+? Life Set Go
This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. Start with an oh energy. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. I.
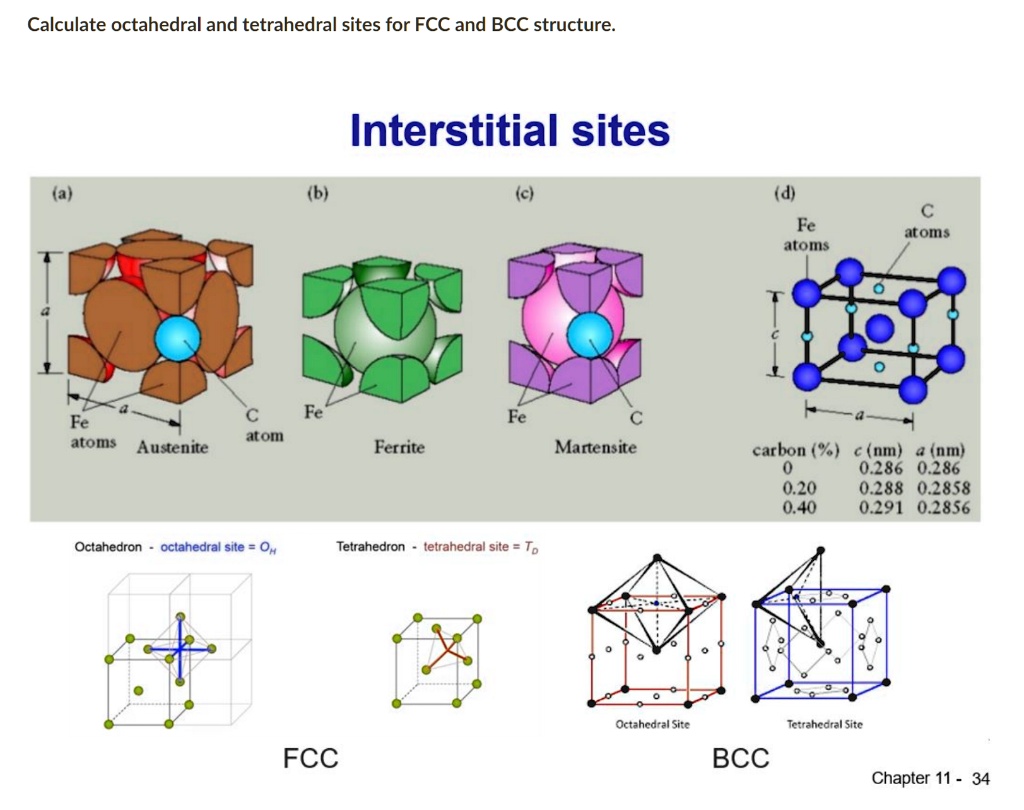
calculate octahedral and tetrahedral sites for fcc and bcc structure
Start with an oh energy. I understand that fe(ii) has 6. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and.
Preparation and Comparative Evaluation of Fe+2/Fe+3 and Mg+2/Fe+3 LDHs
Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Start with an oh energy. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I.
Octahedral (Fe tot þ Mn þ TiÀAl VI ) vs. octahedral (MgLi) diagram
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I understand that fe(ii) has 6. Start with an oh energy. Both fe 2+ and fe 3+ ions give octahedral cyano anionic.
Diagram Fe 2+/ Fe 2+ + Fe 3+ vs Mn/(Mn+Mg) showing comparison between
Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Start with an oh energy. I understand that fe(ii) has 6. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Both fe 2+ and fe 3+ ions give octahedral cyano anionic.
Distortions for a 3d 2 configuration yielding the transition from
Start with an oh energy. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. I.
Fig S9 Schematic electronic DOS of Fe 3+ in octahedral O h and
This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. Start with an oh energy. Now combine the tm dao and ligand fo components to form the energy level diagram for a c4v tm complex. I.
Now Combine The Tm Dao And Ligand Fo Components To Form The Energy Level Diagram For A C4V Tm Complex.
This structure is called a spinel, with $\ce{fe^{2+}}$ ion in tetrahedral coordination and $\ce{fe^{3+}}$> ions in octahedral sites. Start with an oh energy. Both fe 2+ and fe 3+ ions give octahedral cyano anionic complex ions with cyanide ions. I understand that fe(ii) has 6.